Need Help?
Ask an expert
1. How to collect Salivary DHEA-S
APPROVED SALIVARY DHEA-S COLLECTION METHODS
Salivary DHEA-S Collection Protocol
Collection volume, general considerations, and basic guidelines to maximize salivary DHEA-S sample integrity. Use this analyte-specific collection protocol to plan you collection methodology and sampling schemes.

2. How to Assay for Salivary DHEA-S
Send Saliva Samples to Salimetrics
Add to StudyEasy and accurate results from the most trusted Salivary Bioscience Laboratory.
All Lab ServicesOrder Code5130
Salivary DHEA-S ELISA Kit
Add to Study
Salimetrics Assay #1-1252
The Salimetrics Salivary DHEA-S (DHEA-Sulfate) Enzyme Immunoassay Kit was specifically designed to standardize the detection of DHEA-S in saliva samples for research and biomedical laboratories. Using a small sample volume, this assay kit has an extended range which spans the expected DHEA-S levels found in human saliva. The average inter- and intra-assay precision coefficients of variation are low with no deleterious matrix effects often found in saliva which are characterized through dilution- and spike-recovery validation procedures. This DHEA-S assay kit has also been formatted to minimize cross reactivity for related steroids. Read More...| Assay Protocol |
|---|
| Rev. 09.09.22
|
| Specifications | |
|---|---|
| Catalog#: | 1-1252 |
| Regulatory Status: | RUO |
| Format: | 96-well plate |
| Assay Time: | ~ 2 hrs |
| Sample Volume/Test: | 100 µL |
| Sensitivity: | 95.14 pg/mL |
| Assay Range: | 188.9 pg/mL- 15,300 pg/mL |
| Storage Requirements: | 2-8°C |
| Tests Per Kit | |
|---|---|
| Singlet: | 76 |
| Duplicate: | 38 |
| Target Analyte |
|---|
Technical Documentation
Assay Kit Overview
Intended Use
The Salimetrics DHEA-S Enzyme Immunoassay Kit is a competitive immunoassay specifically designed and validated for the quantitative measurement of salivary DHEA-S. It is not intended for diagnostic use. It is intended only for research use in humans and some animals. Salimetrics has not validated this kit for serum or plasma samples.
Introduction
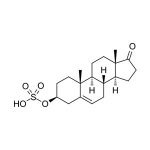
Dehydroepiandrosterone-sulfate (DHEA-S) is a steroid hormone produced primarily in the adrenal cortex. DHEA-S is the most abundant steroid hormone in humans, with circulating concentrations approximately 250 and 500 times higher than those of its unsulfated analog, DHEA, in women and men, respectively. DHEA-S serves as a precursor molecule that is circulated to various target tissues in the body. In those locations the sulfate is removed to yield DHEA, and the DHEA is then further metabolized into various estrogenic and androgenic compounds. DHEA-S is not bound by sex hormone binding globulin (SHBG) in the blood stream and is readily available for conversion to other compounds. Unlike DHEA, DHEA-S does not normally exhibit any diurnal pattern of secretion. Levels of DHEA-S peak around the age of 20 to 30, and then decline to only 20-30% of peak levels by the age of 70 to 80. Critical illness and emotional or physical stress can also cause DHEA-S levels to decline. Lowered DHEA-S levels have been linked with a variety of medical conditions. DHEA-S and DHEA are also synthesized directly in the central nervous system, where they appear to help protect nervous tissue against harmful agents. Studies have begun to explore possible relationships between DHEA-S levels and changes in neurological function, including sense of well-being, cognition, depression, and various other psychiatric disorders. DHEA-S is not lipid soluble, and it cannot enter saliva by passive diffusion through cell membranes like most of the other steroid hormones. Instead, it enters saliva only by squeezing through the tight junctions between cells in the saliva glands, and it is too large to do this readily. It is therefore present in relatively small amounts. Binding proteins or enzymes in saliva that would affect the measurement of free DHEA-S appear largely to be absent. DHEA-S levels measured in whole saliva may be inaccurate if contamination by plasma exudates from the gums or from small injuries in the mouth is present. Subjects should be carefully screened for periodontal disease and advised about proper collection procedures. Saliva may also be screened for blood contamination using the Salimetrics Blood Contamination EIA Kit (Item. No. 1-1302). DHEA-S concentrations in saliva decrease markedly as flow rates increase.
Salivary DHEA-S Assay Principle
This is a competitive immunoassay kit. DHEA-S in standards and samples compete with DHEA-S conjugated to horseradish peroxidase for the antibody binding sites on a microtitre plate. After incubation, unbound components are washed away. Bound DHEA-S Enzyme Conjugate is measured by the reaction of the horseradish peroxidase enzyme to the substrate tetramethylbenzidine (TMB). This reaction produces a blue color. A yellow color is formed after stopping the reaction with an acidic solution. The optical density is read on a standard plate reader at 450 nm. The amount of DHEA-S Enzyme Conjugate detected is inversely proportional to the amount of DHEA-S present in the sample.
3. Technical Summary
| Analyte Summary | |
|---|---|
| Analyte: | DHEA-S |
| Aliases: | Dehydroepiandrosterone sulfate, DHEA sulfate |
| Serum-Saliva Correlation: | NA |
| *Optimum Collection Volume: | 225 μL |
| Special Considerations |
|---|
| DHEA-S is Flow Rate Dependent (pg/mL) |
| Assay Summary | |
|---|---|
| Methodology: | ELISA |
| Sensitivity: | 95.14 pg/mL |
| Assay Range: | 188.9 pg/mL- 15,300 pg/mL |
| Assay Type: | Quantitative |
Background
DHEA-S is a steroid hormone produced primarily in the adrenal cortex. It is the sulfated version of the human steroid DHEA, and, like DHEA, it is secreted in response to ACTH. DHEA-S has been reported to have a diurnal rhythm, but the findings have varied, and some studies found no variation. (1) DHEA-S in the blood stream has a longer half-life, slower clearance, and is more strongly bound to albumin than DHEA, which may affect synchronicity with the DHEA rhythm. (2,3,4) DHEA-S appears to serve largely as a precursor molecule that is circulated to various target tissues in the body. In those locations, the sulfate is removed to yield DHEA, and the DHEA is then further metabolized into various estrogenic and androgenic compounds. This process allows androgens and estrogens to be delivered to the appropriate tissues without leakage of significant amounts into the circulation. (5,6) DHEA-S is also synthesized directly in the central nervous system, where it is thought to help protect nervous tissues against harmful agents. (7,8) DHEA-S has been investigated for relationships to mental and physical stress and psychological and behavioral disorders. ( 9,10,11,12,13) DHEA-S is a charged molecule, and it cannot diffuse through the neutral lipid membranes of the salivary cells like the other neutral steroids. The exact mode of entry into saliva is not known. Formerly, it was thought that DHEA-S enters saliva only by squeezing through the tight junctions between cells, and since it is too large to do this readily only small amounts would be present in saliva. (14) More recent work has identified a large family of organic anion transport polypeptides (OATP) that actively transport molecules such as DHEA-S across membranes. It is therefore seems likely that such a mode of entry occurs for DHEA-S into the saliva glands. (15,16) Salivary levels of DHEA-S are quite low–less than 0.1 % of plasma levels in parotid saliva. (14) However, because levels of DHEA-S in blood are 250 and 500 times higher than DHEA in women and men, respectively, (2) the levels found in saliva are high enough to be measurable. Due to the restrictive mode of entry for DHEA-S into saliva, its levels in saliva decrease as salivary flow rates increase. (14) DHEA-S measurements in saliva must therefore be corrected for flow rate. Because of the much higher levels of DHEA-S in blood, it is important to minimize the risk of blood contamination in the saliva samples. Salivary and plasma levels of DHEA-S show a significant positive correlation. (17)
References & Salivary DHEA-S Research
-
- Summarized in Whetzel, C.A., Klein, L.C. Measuring DHEA-S in saliva: Time of day differences and positive correlations between two different types of collection methods. BMC Res Notes, 3:204.
- Krobath, P.D., Salek, F.S., Pittenger, A.L. et al. (1999). DHEA and DHEA-S: A review. J Clin Pharmacol 39(4), 327-48.
- Rosenfeld, R.S., Rosenberg, B.J., Fukushima, D.K., Hellman, L. (1975). 24-Hour secretory pattern of dehydroisoandrosterone and dehydroisoandrosterone sulfate. J Clin Endocrinol Metab, 40(5), 850-55.
- Carlström, K., Karlsson, R., Von Schoultz, B. (2002). Diurnal rhythm and effects of oral contraceptives on serum dehydroepiandrosterone sulfate (DHEAS) are related to alterations in serum albumin rather than to changes in adrenocortical steroid secretion. Scan J Clin Lab Invest, 62(5), 361-68.
- Labrie, F., Bélanger, A., Cusan, L., Candas, B. (1997). Physiological changes in dehydroepiandrosterone are not reflected by serum levels of active androgens and estrogens but of their metabolites: Intracrinology. J Clin Endocrinol Metab, 82(8), 2403-9.
- Labrie, F., Luu-The, V. Bélanger, A., et al. (2005). Is dehydroepiandrosterone a hormone? J Endocrinol, 187, 169-96.
- Charalampopoulos, I., Alexaki, V.-I., Tsatsanis, C., et al. (2006). Neurosteroids as endogenous inhibitors of neuronal cell apoptosis in aging. Ann N Y Acad Science, 1088, 138-52.
- Baulieu, E.-E., Robel, P. (1998). Dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) as neuroactive neurosteroids. Proc Natl Acad Sci U S A, 95(8), 4089-91.)
- Kellner, M., Muhtz, C., Peter, F., et al. (2010). Increased DHEA and DHEA-S plasma levels in patients with post-traumatic stress disorder and a history of childhood abuse. J Psychiatr Res, 44(4), 215-9.
- Golubchik, P., Mozes, T., Maayan, R., Weizman, A. (2009). Neurosteroid blood levels in delinquent adolescent boys with conduct disorder. Eur Neuropsychopharmacol, 19(1), 49-52.
-
- Azurmendi, A., Braza, F., Garcia, F. et al. (2006). Aggression, dominance and affiliation: Their relationships with androgen levels and intelligence in 5-year-old children. Horm Behav, 50(1), 132-40.
- MacLaughlin, B.W., Wang, D., Noone, A.-M., et al. (2010). Stress biomarkers in medical students participating in a mind body medicine skills program. eCAM, doi:10.1093/ecam/neq039.
- Wang, J.-S., Chen, S.-M., Lee, S.-P., et al. 2009). Dehydroepiandrosterone sulfate linked to physiologic response against hot spring immersion. Steroids, 74(12), 945-49.
- Vining, R.F., McGinley, R.A., Symons, R.G. (1983). Hormones in saliva: Mode of entry and consequent implications for clinical interpretation. Clin Chem, 29(10), 1752-56.
- Konttinen, Y.T., Stegaev, V., Mackiewicz, Z., et al. (2010). Salivary glands — ‘an unisex organ’? Oral Dis, 16(7), 577-85.
- Pomari, E., Nardi, A., Fiore, C., et al. (2009). Transcriptional control of human organic anion transporting polypeptide 2B1 gene. J Steroid Biochem Mol Biol, 115(3-5), 146-52.
- Jezova, D., Hlavacova, N. (2008). Endocrine factors in stress and psychiatric disorders: Focus on anxiety and salivary steroids. Ann N Y Acad Sci, 1148, 495-503.
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)